New research supported by the National Institutes of Health delineates how two relatively common variations in a gene called KIF3A are responsible for an impaired skin barrier that allows increased water loss from the skin, promoting the development of atopic dermatitis, commonly known as eczema. This finding could lead to genetic tests that empower parents and physicians to take steps to potentially protect vulnerable infants from developing atopic dermatitis and additional allergic diseases.
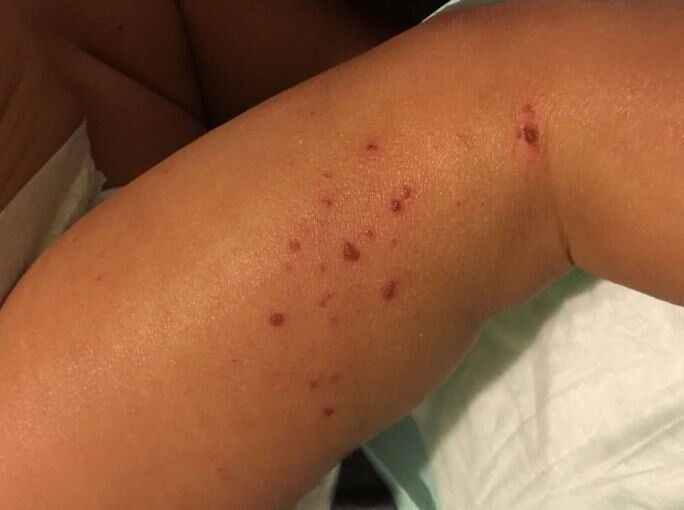
Atopic dermatitis is an inflammatory skin condition that affects up to 20% of children in developed countries. This chronic disease is characterized by dry, thickened and intensely itchy skin, particularly in skin folds. People with eczema are more susceptible to bacterial, viral and fungal skin infections and frequently develop additional allergic diseases such as asthma.
KIF3A is a gene that codes for a protein involved in generating signals from the outside to the inside of a cell, part of a complex sensory apparatus. Previously, scientists had identified an association between two genetic variations in KIF3A and asthma in children who also had eczema. In the new study, the researchers found that these variations, or single nucleotide polymorphisms (SNPs), changed parts of the KIF3A gene to a form that can regulate, through a process called methylation, the rate at which a gene is transcribed into the blueprint for protein production. The investigators confirmed that skin and nasal-lining cells from people with the KIF3A SNP variants had more methylation and contained fewer blueprints for the KIF3A protein than cells in which KIF3A lacked the SNPs. In addition, the researchers demonstrated that people with the SNP-created regulating sites had higher levels of water loss from the skin.