Today, we have to share some bad news about Organovo and its liver tissue bioprinting program.


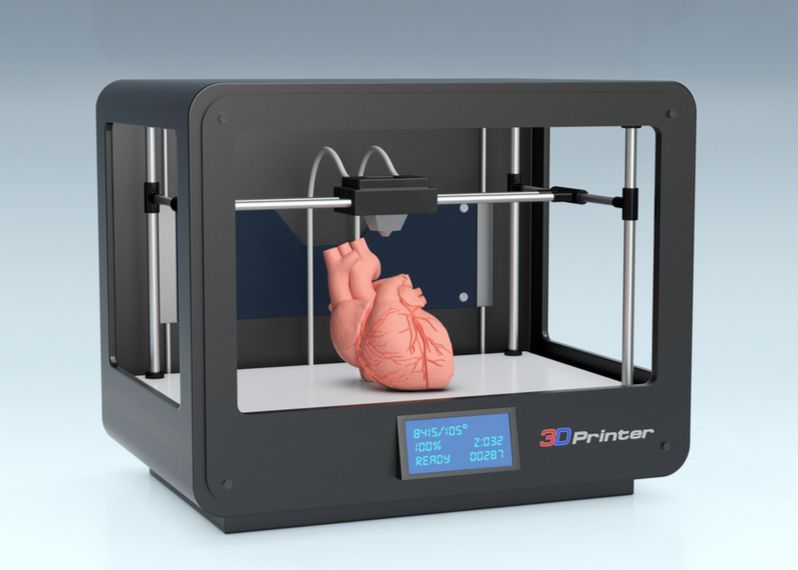
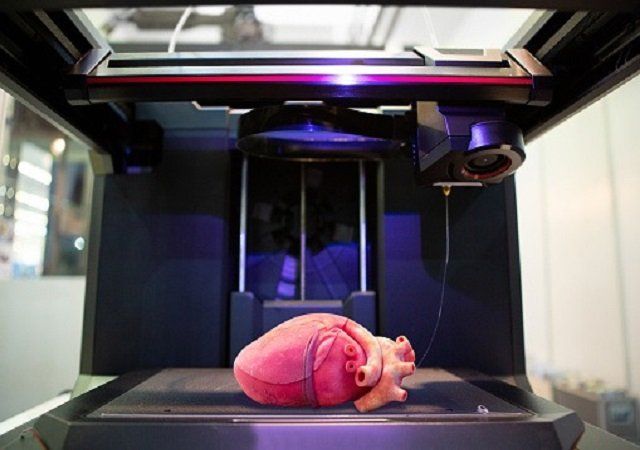
While in its early stages, bioprinting of human tissue is an emerging technology that is opening up some exciting possibilities, including the potential to one day 3D print entire human organs. This scientific objective has now grown a little bit closer, with researchers at Carnegie Mellon University reporting a breakthrough that enabled the printing of full-scale heart components that in some cases functioned similarly to the real thing.
Over 4000 patients in the United States alone are waiting for a heart transplant, while millions of others worldwide need hearts but are ineligible for the waitlist. The need for replacement organs is immense, and new approaches are needed to engineer artificial organs that are capable of repairing, supplementing, or replacing long-term organ function.
A team of researchers from Carnegie Mellon University has published a paper in Science that details a new technique allowing anyone to 3D bioprint tissue scaffolds out of collagen, the major structural protein in the human body. This first-of-its-kind method brings the field of tissue engineering one step closer to being able to 3D print a full-sized, adult human heart.
The technique, known as Freeform Reversible Embedding of Suspended Hydrogels (FRESH), has allowed the researchers to overcome many challenges associated with existing 3D bioprinting methods, and to achieve unprecedented resolution and fidelity using soft and living materials.
Each of the organs in the human body, such as the heart, is built from specialized cells that are held together by a biological scaffold called the extracellular matrix (ECM). This network of ECM proteins provides the structure and biochemical signals that cells need to carry out their normal function. However, until now it has not been possible to rebuild this complex ECM architecture using traditional biofabrication methods.


Kyle Reese: The Terminator’s an infiltration unit, part man, part machine. Underneath, it’s a hyperalloy combat chassis — micro processor-controlled, fully armored. Very tough. But outside, it’s living human tissue — flesh, skin, hair, blood, grown for the cyborgs…
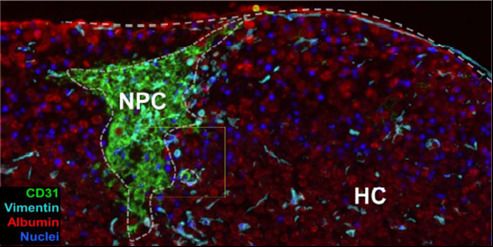
3D bioprinting is the automated fabrication of multicellular tissue via spatially defined deposition of cells. The ability to spatially control deposition in the x, y and z axes allows for creation of tissue-specific patterns or compartments, with in vivo-like architecture that mimics key aspects of native biology.
3D bioprinted tissues exhibit a microenvironment more suited to in vivo-like cellular function in comparison to traditional 2D monoculture (or monolayer co-cultures), as well as maintenance of a more defined architecture than is observed in self-aggregated co-culture models.
“One of the biggest roadblocks to generating functional tissue replacements has been our inability to print the complex vasculature that can supply nutrients to densely populated tissues,” said Jordan Miller, assistant professor at Rice University in the US.
“Further, our organs actually contain independent vascular networks — like the airways and blood vessels of the lung or the bile ducts and blood vessels in the liver,” Miller said.
“These interpenetrating networks are physically and biochemically entangled, and the architecture itself is intimately related to tissue function. Ours is the first bioprinting technology that addresses the challenge of multi vascularisation in a direct and comprehensive way,” he said.