Tag: Neurobiology


The Alzheimer’s Hypothesis
The Alzheimer’s Hypothesis
Introduction
Alzheimer’s disease was first discovered in 1907 in a 51 year old woman by the German physician A. Alzheimer. One of the first changes noticed was an eruption of jealous feelings towards her husband. It wasn’t long before symptoms of rapid memory impairment were observed. The impairments prevented her from finding her way out of her home, She hid herself, she would drag objects to and fro, and occasionally screamed because she believed people were out to kill her.
When she was institutionalized her gestures would show a complete helplessness. As common in most Alzheimer’s patients, she was disoriented as to time and place. At times she would state that she didn’t understand anything, felt confused, and totally lost. When the doctor came in to see her she would consider it as an official visit and would apologize for not having finished her work. Other times she would be terrified and start to yell that the doctor wanted to operate on her. Other times she would send him away in complete indignation uttering phrases indicating that she was afraid that the doctor wanted to damage her woman’s honor. At times she would become completely delirious , dragging her blankets and to and fro, calling for her husband and daughter, and seeming to experience auditory hallucinations. She would often scream for hours and hours in a horrible voice. Mental regression advanced quite steadily. After four and a half years of illness the patient finally died.
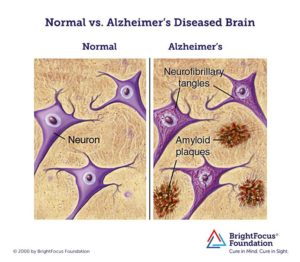
Alzheimer performed a postmortem examination of the woman’s brain. He paid special attention to changes in the “neurofibrils,” fibers in the cytoplasm of a nerve axon — elements of the cytoskeleton that can be stained by a silver solution.
The Bielschowsky silver preparation showed very characteristic changes in the neurofibrils. Looking inside an apparently normal-looking cell, however, one or more single fibers became observably prominent through their striking thickness and specific impregnability. At a more advanced stage many many fibers arranged in parallel showed the same changes. Over time they formed dense bundles and gradually advanced to the surface of the cell. The cell and nucleus would eventually disappear, leaving only a tangled bindle of fibrils where the neuron once was.
Since these fibrils can be readily stained with certain dyes, a chemical transformation of the fibril substance must have taken place. This might be why the fibrils survived the destruction of the cell. The transformation of the fibrils seems to go hand in hand with the storage of an as-of-yet not closely investigated pathological byproduct of neuronal metabolism. Such alterations were found in about one-quarter to one-third of all the neurons in the cerebral cortex. Numerous neurons, especially in the upper cell layers, had completely disappeared.
The number and distribution of neurofibrillary tangles, what are commonly known as the “tombstones” of dead and dying neurons, are correlated with the severity of the symptoms in Alzheimer’s disease.
Phase1
What is the purpose of beta-amyloid?
Alzheimer’s disease causes impairment with memory, thinking, and behavior. Alzheimer’s affects 5.5 million people age 65 and older and approximately 200,000 individuals under the age 65. Alzheimer’s disease is the 5th leading cause of death in the United States. The root cause of Alzheimer’s is still unknown. There are several hypothesis that attempt to explain it. This article focuses on the research being done in a joint effort between a team from Harvard University being led by Rudolf Tanzi and Robert Moir at Boston Massachusetts General Hospital.
Tanzi and Moir investigated the development of Alzheimer’s disease in animals and found the development of amyloid plaques is not unique to humans. 70 percent of vertebrates share certain sequences of particular amino acids in human beta-amyloid. Moir believes because this sequence is shared so widely and has not changed over time that it must be doing something very important. Moir discovered that the antimicrobial peptide LL37 acts as one of the brain’s defenses against microbes alongside beta-amyloid.
In order to test this hypothesis, the team injected the brains of mice that were bred to develop Alzheimer’s plaques comparable to the way humans do with bacteria. Overnight, the mice produced plaques. Tanzi discovered that each plaque had a single bacterium in it. A single bacterium can induce an entire plaque.
25 May 2016
Science Translational Medicine 25 May 2016:
Vol. 8, Issue 340, pp. 340ra72
DOI: 10.1126/scitranslmed.aaf1059
Amyloid-β peptide protects against microbial infection in mouse and worm models of Alzheimer’s disease
Phase 2
The Role of Sleep on the Brian’s Filtration System
Lulu Xie and team recently uncovered one of the key functions of sleep. The team compared 2 arousal states in the mouse, sleeping and awake, using state-of-the-art in vivo two-photon imaging. The researchers found that metabolic waste products of neural activity were cleared out of the sleeping brain at a faster rate than during the awake state. Like the accumulation of beta amyloid plaques, sleep is a cross-species phenomenon.
The study by Xie et. al reports that the critical function of metabolic homeostasis is dependant on sleep. The team utilized real-time assessments of tetramethylammonium diffusion and two-photon imaging in live mice. The experiment found a 60 percent increase in the interstitial space in association of natural sleep or anesthesia. This resulted in a noticeable convective exchange of cerebrospinal fluid and interstitial fluid, which are found in the spaces between the blood vessels and surrounding cells.
Thus as a result convective fluxes the rate of β-amyloid clearance during sleep. Hence, the restorative function of sleep may be a consequence of the enhanced removal of potentially neurotoxic waste products that accumulate during our waking hours.
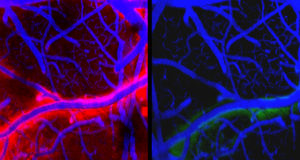
Sleep Drives Metabolite Clearance from the Adult Brain
Science 18 Oct 2013:
Vol. 342, Issue 6156, pp. 373–377
DOI: 10.1126/science.1241224
In a separate study Ehsan Shokri-Kojori and team published a study on April 9 2018 showing that just 1 night of sleep deprivation builds β-Amyloid within the brain.
PNAS April 24, 2018 115 (17) 4483–4488

On September 5th of this year The Sleep Research Society came out with a report that collaborates with the data suggesting just how important the role of sleep is to prevent Alzheimer’s disease. The Baltimore Longitudinal Study of Aging, conducted by Adam P Spira et al showed that those who experienced excessive sleepiness during normal waking hours were nearly three times more likely to have brain deposits of beta amyloid than those who didn’t.
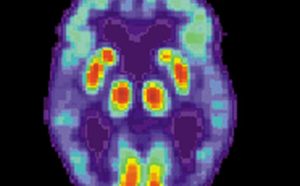
PET scan of a human brain with Alzheimer’s disease
Sleep, zsy152
Published: 05 September 2018
The accumulated evidence above shows that Beta-Amyloid are part of the brain’s ancient immune system . The evidence also indicates that the buildup of Beta-amyloid plaques occurs when it’s not flushed out. Many researchers, including Rudolf Tanzi and Robert Moir believe that it is the plaques that cause the damage to the brain.
Phase 3
The fIltration System of the Brain
The re-discovery of cellular membranes around the brain known as meningers has a network of lymphatic vessels that drain macromolecules from the CNS (cerebrospinal and interstitial fluids) into the cervical lymph nodes in mice. This lymphatic system was first discovered in 1787 (Mascagni, P. Vasorum lymphaticorum corporis humani historia et ichnographia) and was rediscovered by Da Mesquita et al.
Their research showed that meningeal lymphatics play a central role in brain health and disease by helping to maintaining both cognitive function and proteostasis.
The purpose of the lymphatic system in the body is to drain tissue of interstitial fluid that houses cellular debris and toxic chemicals. A rich protein-fluid called lymph is formed by interstitial fluids which circulates through the lymphatic system before returning to the blood. Lymph is filtered through the lymph nodes. Lymph nodes are responsible for initiating immune responses when foreign particles are detected.
Because the brain does not have its own lymphatic vessels it utilizes the interstitial fluid (ISF) along the walls of blood vessels to transport waste and proteins from the parenchyma to reach the cerebrospinal fluid that circulates through the meninges. The transvascular process is the process in which metabolic waste products, proteins, and other molecules in these fluids are removed from the brain by being transported along the walls of blood vessels, which crosses the blood-brain barrier. To what extent, if any, the meningeal lymphatic vessels were also involved in waste clearance is still unknown.
To verify the critical role of the meningeal lymphatic vessels Da Mesquite et. al injected a drug designed to damage the vessels into one of the three principal openings in the subarachnoid space between the arachnoid and pia mater layers of the meninges surrounding the brain, collectively referred to as the cisterna magna. In order to track the CSF they injected a fluorescent tracer molecule. This provided insight as to where the fluid goes in mice lacking meningeal lymphatic vessels. Da Mesquite et. al noted that the the tracer did not travel deep into the cervical lymph nodes in mice lacking meningeal lymphatic vessels. Injecting high concentrations of a tracer into the CSF can cause the diffusion of a tracer into the brain along blood vessels as shown by previous work — but this too was also reduced. The researchers used different tracers, surgically closing off drainage to the deep cervical lymph nodes; and examined mice with impaired lymphatic vessel development . The authors were able to confirm through these various methods the results by doing so.
The authors also noted that deficits within spatial orientation and memory were a result of meningeal lymphatic destruction.
Another interesting finding by the researchers was a change in gene expression within the hippocampus, a substructure crucial to memory and spatial orientation.
The gene expression resembled those previously observed in neurodegenerative disorders. The accumulated evidence by these experiments suggests collectively that the drainage of brain ISF and CSF by meningeal lymphatics is critical to proper function of cognition within the brain.
These findings present an interesting question: where exactly did the tracers go? One study (https://onlinelibrary.wiley.com/doi/abs/10.1038/icb.1951.30) suggests that tracers injected into the cisterna magna are transported primarily in the blood and that the lymphatic system is only secondary. To reveal whether the impairment of meningeal lymphatics can lead to a shift in the pathways responsible for controlling brain proteostasis the simultaneous measurements of tracers movements meningeal lymphatics, other lymphatic vessels, (for example the neck) and the blood may give insights into whether there’s an increase in the transvascular removal of waste products across the blood-brain barrier and/or their drainage into the venous system in the meninges.
The next observation by Da Mesquita’s team was a decrease in the diameter and coverage of meningeal lymphatic vessels through induced aging. The researchers also observed a decrease in drainage of tracers from the ISF and CSF into deep cervical lymph nodes. The signaling of the pathway involving vascular endothelial growth factor C (VEGF-C) and its receptor VEGFR3 promotes lymphatic vessel growth in mice. Likewise when the pathway involving endothelial growth was damaged it caused a loss of meningeal lymphatic vessels. Moreover the authors showed that there was an increase diameter of meningeal lymphatic vessels by providing treatment VEGF-C, improving lymphatic drainage as a result. Consistent with the accumulated research, Da Mesquita et al. showed that the drainage of CSF tracer could be restored into deep cervical lymph nodes by the local delivery of the vegf-c into the cisterna of old mice with a viral vector . This resulted in the restoration of spatial orientation.
The accumulation of amyloid-β protein within the brain, a hallmark of Alzheimer’s disease, causes age related impairments in the transvascular clearance of waste. The effects of of ablating the meningeal lymphatics in two mouse models of Alzheimer’s disease, in which amyloid-β protein is produced in neurons and secreted into the ISF was investigated by Da Mesquita and team. The results showed that ablation led to the accumulation of amyloid-β protein in the meningenges. The authors also noted to the potential relevance for humans by showing that amyloid-β had accumulated in meningenges of people who had Alzheimer’s disease as well.
When amyloid-β protein deposition in the brain parenchyma first became apparent, Da Mesquita and colleagues discovered that the two mouse models did not exhibit any apparent structural or functional changes in the meningeal lymphatics at the time. Cognitive impairment could not be prevented in either model using viral delivery vegf-c, which points to another disruption in another clearance pathway the early amyloid-β deposition and cognitive impairments in these two models-most likely the transvascular clearance. More than likely an increased burden is placed upon meningeal lymphatic system due to the gradual deterioration of transvascular-clearance routes through aging. Faulty lymphatic drainage of amyloid-β and other proteins from the ISF and CSF may be caused when the capacity of the lymphatic system is reached. This gives strong evidence to suggest that there is a very delicate relationship between meningeal lymphatics and blood vessels in the regulation of proteostasis in the brain.
In order to open new directions for research into cognition, neurodegeneration, and Alzheimer’s disease, the improvement of our understanding of waste clearance pathways from the brain, how the ISF and the CSF drain into the meningeal lymphatics, and how these lymphatic vessels interact with the blood vessels at the blood-brain barrier is vital. The potential of improving clearance with meningeal lymphatics to rebuild brain proteostasis, and may lessen amyloid-β protein deposition have been shown by Da Mesquita et al. to rely upon the local growth of lymphatic vessels. Whether or not the improvement of the impaired function of blood vessels due to aging, and whether enhancing clearance at the blood-brain barrier can improve lymphatic drainage function, remains to be to determined.
Nature 25 July 2018
A lymphatic-waste disposal system implicated in Alzheimer’s Disease
Naturevolume 560, pages185–191 (2018
Functional aspects of meningeal lymphatics in ageing and Alzheimer’s disease
The accumulated evidence shows that Beta-amyloid may prove to be beneficial in killing off microbes, but also that proper rest is needed to activate the filtration system of the brain to consistently flush out these neurotoxins before they cause impairment or disease . Further research into the prevention of age-related damage to the brain’s lymphatic system may offer clues as to how we can prevent cognitive decline of age-related diseases. Ideally this will be done with protective mechanisms the brain already has in place.
Journal of Alzheimer’s Disease — Volume Pre-press, issue Pre-press, vol. 65, no. 2, pp. 659–682, 2018
Accepted 5 July 2018
Published: 21 August 2018
Postsynaptic Proteome of Non-Demented Individuals with Alzheimer’s Disease Neuropathology
Further analysis of these key factors could pave the road to ending Alzheimer’s and other age-related diseases.
Nicholi Avery, [email protected]
Adam Alonzi, [email protected]
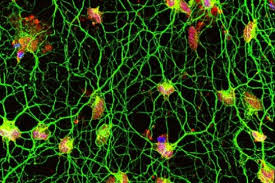
Experimental Drug Injection Causes the Brain to Grow New Neurons
For the first time ever researchers have had a breakthrough in creating a cocktail of drugs that caused new neurons to grow in the brains of mice.
In my last article I gave a detailed account on the debate of neurogenesis. While some neuroscientists claim that neurogenesis takes place within the adult mammalian human brain other researchers contest that idea claiming that new neurons stop developing at a very young age. Whichever side of the debate you are on one thing remains certain, that there are neurological diseases that leave negative impacts on cognitive function. This has left researchers looking for various ways to treat Alzheimer’s, Parkinson’s, and other brain damage.
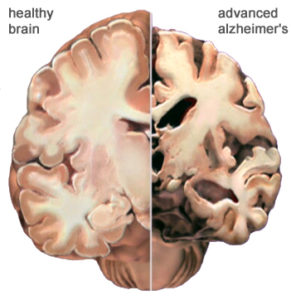
While the brain is incredibly complex and past research has failed to offer much hope for Alzheimer’s disease, Hongkui Deng at Paking University Health Science Center in China may finally be able to change that.
For the first time ever researchers have had a breakthrough in creating a cocktail of drugs that caused new neurons to grow in the brains of mice.
What this cocktail does when injected into the brain is it hijacks the astrocytes into behaving like new neurons. This is significant for a number of reasons. One important detail about astrocyte cells is that they can survive after a stroke while regular neurons die. Another important detail is that there are 10 times more astrocytes in the brain than neurons. That means that there are 1 trillion glia cells within the brain. So not only are they more resilient than neurons they outnumber them too.
Deng and his team of researchers have found that when the cocktail is injected into the brains of mice that it effectively gives the cell a new identity by erasing its old one and giving it a new one. Not only did the cells change shape but they showed change in gene activity too.
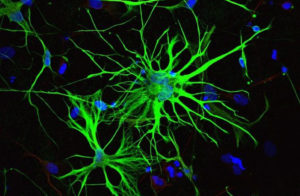
The results were substantial. While it remains speculative as to how closely the cells resemble normal neurons, around 80 to 90 per cent of the astrocytes started to resemble neurons and even mimicked their behavior by electrical signals the same way regular neurons do.
“They are unlikely to be a 100 percent match” Deng stated. “But the treatment was safe and none of the mice developed any health problems”.
Matthew Grubb at King’s College London states that “if it holds up it’s absolutely amazing, and has a lot of potential applications and exciting consequences. If you’ve got a degenerating brain, for example in Alzheimer’s disease, and you could get the brain to regrow neurons itself, it would be a huge step forward.”
The next step for the researchers will be to test the cocktail in mice that have had a stroke. The hope is that the cocktail will cause the astrocytes to behave as neurons and help the mice recover.
While Grubb admits it is difficult to predict the effects of the treatment in humans, if it works in mice then it offers new hope for those who suffer from neurodegenerative diseases such as Alzheimer’s or Parkinson’s. While it is unlikely to bring back lost memories Grubb thinks it might restore the ability to create new ones.
Even though the research is promising there still remain challenges as Roger Barker at the University of Cambridge points out. The sheer numbers of cells lost in a neurodegenerative disease is something to consider. “In Parkinson’s, a quarter of a million cells are lost from either side of the brain. Currently Barker is conducting clinical trials of implants of brain tissue taken from aborted fetuses as a treatment for Parkinson’s.
Another challenge is to distinguish the types of neurons the drugs will make. As Grubb points out, if you make too many of the type of neurons that excites their neighbors you end up triggering epilepsy. Grubb also points out that different brain disorders effects different types of neurons which is another reason why we need to be capable of distinguishing them. The neurons that die from Parkinson’s are the neurons that create the chemical dopamine for example.
Another example is balancing the risks for rewards. As Grubb points out “you’d have to have extremely good control over what cells you’re programming, where they’re going to go, and which cells they’ll connect to. If the treatment were to be used to boost grey matter, for example, this could provide a way to improve skills like memory. However, too much grey matter has been linked to causing people to be being easily distracted.
While the research is definitely groundbreaking it is still in its early stages. Hopefully in the near future it will offer treatments to those who need them.
The Neurogenesis Debate
The Neurogenesis Debate
Written by Nicholi Avery
On March 7 2018, a study was published in the highly esteemed journal Nature by an international team of scientists claiming that #neurogenesis starts to rapidly decline in the #human brain as early as 13 years old and becomes undetectable in adults. This rocked the scientific community as there has been a long-established theory that neurogenesis takes place throughout the course of life in the mammalian #brain. Until the 1990s, neurologists were practicing their profession under the doctrine established in the late 19th to early 20th century by the prominent histologist Ramon y Cajal, often referred to as a god of neuroscience;
“Once the development was ended, the founts of growth and regeneration of the axons and dendrites dried up irrevocably. In the adult centers, the nerve paths are something fixed, ended, and immutable. Everything may die, nothing may be regenerated. It is for the science of the future to change, if possible, this harsh decree.”
It wasn’t until later between the 1960s and the 1990s that possible theories for neurogenesis started to develop.
Neurogenesis
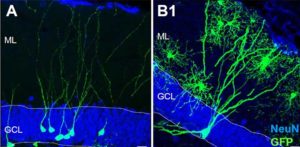
What is neurogenesis exactly?
Neurogenesis is the process by which nerve #cells, known as #neurons, are produced by #neural stem cells (NSC), and it occurs in all animals. This growth is primarily believed to take place in the hippocampus. Despite skeptics, this has been witnessed in a number of vertebrates, and it continues in the mammalian brain throughout life.
Traditionally neurogenesis was believed to only take place during the embryonic and perinatal stages in #mammals. In June 1965 associate professor at MIT Joseph Altman provided ground-breaking research providing histological evidence of postnatal hippocampal neurogenesis in rats. Altman’s research showed that young rats injected with thymidine‐H3 labeled cells declined rapidly with increased age at the time of injection. The researchers write: “The number of labeled cells declined rapidly with increased age at the time of injection. Histological studies showed the presence in young rats of a large germinal matrix of mitotic cells in the ependymal and subependymal layers of the third and lateral ventricles.
The areal extent and cell population of this germinal pool declined rapidly from birth on, with a transient rise with a peak at about 15 days. During this latter period, the number of “undifferentiated” cells near the granular layer of the dentate gyrus showed a rapid rise with a subsequent decline. The decline in the number of “undifferentiated” cells was accompanied by a rise in the number of differentiated granule cells. Cell counts in homologous parts of the dentate gyrus indicated a six‐fold increase in the number of differentiated granule cells from birth to three months.”
Though Altman’s research was published in highly esteemed journals, it was not readily accepted by leaders in the field because as Sapolsky notes in ‘Behave: The Biology of Humans at our Best and Worst’, Altman did not get tenure. Within a few years he lost his funding for his neurogenesis work.
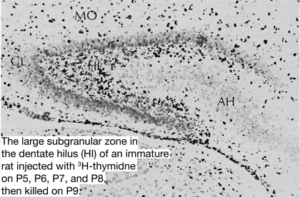
A decade later Michael Kaplan, assistant professor at the University of New Mexico, presented more evidence for neurogenesis utilizing new techniques, also encountering the same problems as Altman did and so, once again, it was rejected by senior figures in the field. The most crushing blow to the neurogenesis debate came from Pasko Rakic who presently works in the Yale School of Medicine Department of Neuroscience in New Haven, Connecticut. Rakic publicly criticized Kaplan’s work and denounced it. This had a devastating effect on Kaplan’s career. He left the field and pursued a career in medicine.
The question as to whether neurogenesis took place in the adult mammalian brain remained an open one and any discussion about neurogenesis was put on hold for another 15 years.
Another unexpected breakthrough in the evidence for neurogenesis took place by a highly esteemed and accomplished neuroscientist named Fernando Nottebohm. A study conducted by Paton JA and Nottebohm FN in 1984 successfully demonstrated the functional integration of new neurons in the adult central nervous system. They demonstrated this by injecting adult canaries with 3H-labeled thymidine, a marker of DNA synthesis. Once the birds matured, it was shown that neurons were generated during or after the 3H-labeled thymidine treatment and subsequently incorporated into functional #neural circuits. The critics were silenced by the quality of Fernando’s work.
While this provided evidence that neurogenesis took place within the adult brain of songbirds, what about mammals?
In 1992 Reynolds BA1 and Weiss S. presented data for the generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system. They did this by isolating cellular striatum of the adult mouse brain and were influenced to rapidly grow within a controlled experimental setting by epidermal growth factor. Finally the acceptance of neurogenesis started to gain some traction in the field.
Soon after this research other neuroscientists were applying newer and more advanced methods developed by Elizabeth Gould of Princeton and Fred “Rusty” Gage of Salk Institute. They were quickly discovering new neurons in not only songbirds, but mammals too. Even Rakic began to rethink his position.
As neuroscience blossomed neurogenesis was observed not only in mice but other rodents as well. However, many scientists were not only skeptical as to how many new cells were actually made by neurogenesis and how long they actually lived, but also questioned whether neurogenesis took place in other mammals such as primates. Though Rakic was becoming more convinced by the evidence of neurogenesis he remained skeptical if it actually occurred where it really counts, such as the cortex. As time went on the debate as to whether neurogenesis took place in primates and monkeys was settled as evidence showed that it does.
Yet the answer as to whether neurogenesis took place in the human brain remained elusive.
Dr. Eriksson and team from the Department of Clinical Neuroscience, Institute of Neurology, Sahlgrenska University Hospital, Göteborg , Sweden set out to test what many scientists in the field have been wondering ever since neurogenesis was demonstrated to take place in primates. Does neurogenesis take place in the human brain as well?
During the fall of November 1st in 1998 Eriksson and team published the first peer-reviewed article on whether neurogenesis takes place in the mature human brain.
Eriksson obtained human brain tissue that was postmortem from patients who had been treated with the thymidine analog, bromodeoxyuridine (BrdU), which labels DNA during the S phase. In order to show that new neurons they used immunofluorescent labeling for BrdU and for one of the neuronal markers, NeuN, calbindin or neuron specific enolase (NSE), showing that new neurons, as defined by the markers, are generated from dividing progenitor cells in the dentate gyrus of adult humans. The results were conclusive! The team wrote: “we demonstrate that new neurons, as defined by these markers, are generated from dividing progenitor cells in the dentate gyrus of adult humans. Our results further indicate that the human hippocampus retains its ability to generate neurons throughout life.”
This was a huge success for Eriksson. The loss of neurons was thought to be irreversible and is one of the leading causes for brain disease. The discovery of neurogenesis could offer key insight into how we can offset brain related diseases as well as provide treatment. It also gives insight into one of the most amazing things about our brain.
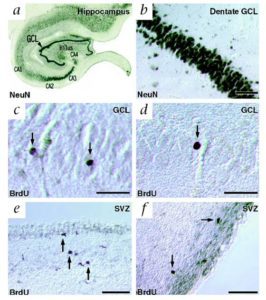
As time went on more evidence was accumulated for neurogenesis taking place in the human brain. Here is a brief timescale of the evidence since Eriksson’s breakthrough research:
1) 2000 Fred “Rusty” Gage PhD showed that neural #stem-cells not only exist in the developing mammalian nervous system but also in the adult nervous system of all mammalian organisms, including humans. Mammalian neural stem cells.
2) 2004 Arturo Alvarez PhD demonstrated that the adult mammalian brain retains neural #stem cells that continually generate new neurons within two restricted regions: the subventricular zone (SVZ) of the lateral ventricle and the dentate gyrus subgranular zone (SGZ) of the hippocampus. Alvarez also discovered the common themes that begin to define adult neurogenic niches. Maintaining Germinal Niches in the Adult Brain.
3) 2006 Pierre-Marie Liedo PhD discovered that the #plasticity of the brain morphological and physiological changes are possible at many levels, including that of the entire cell, even in the face of a varying environments. Adult neurogenesis and functional plasticity in neuronal circuits.
4) 2004 Arturo Alvarez PhD demonstrated that the adult mammalian brain retains neural stem cells that continually generate new neurons within two restricted regions: the subventricular zone (SVZ) of the lateral ventricle and the dentate gyrus subgranular zone (SGZ) of the hippocampus. Alvarez also discovered the common themes that begin to define adult neurogenic niches. How wide spread Is adult neurogenesis in mammals?
5) 2008 Xin Duan, PhD demonstrated the sequential steps of adult neurogenesis, ranging from neural precursor proliferation to synaptic integration of newborn neurons. Development of neural stem cell in the adult brain.
6) 2010 Wei Deng, James B. Aimone & Fred H. Gage explored how the adult hippocampal neurogenesis affects learning and memory making important discoveries. New neurons and new memories: how does adult hippocampal neurogenesis affect learning and memory?
While the evidence for neurogenesis is compelling, the newly published study mentioned above casts doubt on the long established evidence based upon neurogenesis. The study was led by PhD Robert Sapolsky’s graduate student Shawn Sorrells and a team of international scientists.
They document what much of the earlier evidence for neurogenesis seemed to indicate, that the birth of new neuron takes place in the early development of mammalian brain. However Sorrells and team claim to have demonstrated that a defined population of progenitor cells does not coalesce in the subgranular zone during human fetal or postnatal development. They also state that they found that the number of proliferating progenitors and young neurons in the dentate gyrus declines sharply during the first year of life and only a few isolated young neurons are observed by 7 and 13 years of age.
This is a far cry from the decades worth of research presented in favor of neurogenesis.
Does Neurogenesis exist in the human brain? I reached out to world renowned neuroendocrinologist Robert Sapolsky inquiring about this latest study. In an email Sapolsky assured me that they were extremely careful and that he completely believes the data of the study. Sapolsky also stated that the key to this study in comparison to his own work is that it involves human brains (and is the most thorough study of adult human neurogenesis ever done), whereas everything he has done is with rodents. Though I highly respect Doctor Sapolsky, other neuroscientists, are skeptical of such a report as it is the first of its kind.
Dr. Sandrine Thuret supports the long established data of neurogenesis and she believes that the failed replication of one experiment for neurogenesis is not convincing enough to dismiss decades worth of research. Dr. Sandrine Thuret is Principal Investigator & Lecturer in Neural Stem Cell Research at the prestigious King’s college London.
Dr. Saul Villeda in a recent paper demonstrated that the tet2 molecule found in the blood of younger mice plays a vital role in the development of new neurons. Villeda believes that neurogenesis is a signpost of regeneration in the brain, but thinks ultimately that it is changes to the neurons themselves preventing synapse loss, and boosting plasticity that will improve cognition. Villeda is hopeful that a similar molecule may help birth new neurons in the human brain as well.
Does neurogenesis take place in the adult mammalian brain?
One thing is for certain, to break free of old belief systems is not as simple as being exposed to evidence or even proof that the beliefs are wrong.
Even in the scientific community, which prides itself on reason and rationality, we see a long history of proof being ignored for years, decades, even generations. It has been said that science progresses “funeral by funeral” as we wait for the “old guard” to die off for new facts to be accepted.
Scientists “knew” that surgeons using disinfectants and washing their hands before attending to childbirth would prevent infections and save lives. The doctors ignored the evidence and refused to change their practices of going straight from autopsies to surgeries. It took years for this practice of handwashing to finally be accepted.
It requires a willingness to accept the truth after we have discovered it.
Examples like this are numerous and extend to every field of human endeavor.
The debate in the scientific community on neurogenesis demonstrates this all too well.
On April 5th, 2018, the latest study of neurogenesis was published in Cell claiming to once again have demonstrated the growth of new neurons in the hippocampal region of the adult human brain. This particular study is important because the researchers utilized the method of stereology—the gold standard of neurogenesis studies in animals—to count immature and mature neurons in the hippocampi of healthy humans. Stereology can determine the number of individual cell types in tissue, even when the sample has been sliced into sections. Lead researcher Maura Boldrini et al. state: “We found similar numbers of intermediate neural progenitors and thousands of immature neurons in the DG, comparable numbers of glia and mature granule neurons, and equivalent DG volume across ages.
Nevertheless, older individuals have less angiogenesis and neuroplasticity and a smaller quiescent progenitor pool in anterior-mid DG, with no changes in posterior DG. Thus, healthy older subjects without cognitive impairment, neuropsychiatric disease, or treatment display preserved neurogenesis. It is possible that ongoing hippocampal neurogenesis sustains human-specific cognitive function throughout life and that declines may be linked to compromised cognitive-emotional resilience.”
The understanding of neurogenesis is important as it would give scientists data as to how the brain can repair itself which could lead to breakthrough research that could treat brain related diseases or damage such as Alzheimer’s disease.
I shared my article with Dr. Sorrells and he requested that I share the critique of the study in Cell:
In our view, the new study’s exclusive reliance on labeling cellular proteins associated with new neurons is not sufficient evidence to conclude that the cells being observed really are new neurons. Based on the representative images they present, the cells they call new neurons in the adult hippocampus are very different in shape and appearance from what would be considered a young neuron in other species, or what we have observed in humans in young children.
This is why in our study we also performed careful analysis of cell shape and structure using light and electron microscopes, which revealed that similarly labeled cells in our own adult brain samples proved to be neither young neurons nor neural progenitors, but rather non-neuronal glial cells expressing similar molecular markers.
We also have technical concerns about some of the key protein labeling performed in this study. Based on the images presented in the paper, some of these labels appear to be dominated by optical contamination (“bleedthrough”) from other stains used in the same experiments, further calling into question the results overall.
Finally, we also question the assertion by Boldrini et al that the subgranular zone (SGZ), the key site of adult neurogenesis in the rodent brain, is present in their adult human brain samples. None of their histology actually shows a layer of small, frequently proliferating cells and their blood vessel staining in figure 4 fails to show the dense plexus of blood vessels that characterizes the SGZ in rodents and is considered a key structure for neurogenesis in the adult mammalian brain. This is consistent with our own observation that in humans the SGZ never coalesces as a supportive site for adult neurogenesis in the dentate gyrus.
Given the unclear nature of the cells presented, the lack of a clear SGZ, and the developmental trajectory that we describe in our study, with new neurons clearly present in children but not in adults, we think that the simplest interpretation is that neurogenesis in the adult human dentate gyrus is absent or extremely rare. It boils down to interpretation of equivocal cells which we took extra steps to characterize extensively and showed not to be new neurons as they first appeared.
In the future we may need to look at how we can artificially stimulate neurogenesis within the brain or creating artificial neurons.
Resources and citations:
“Human Hippocampal Neurogenesis Persists Throughout Aging.”
Cell Volume 22, Issue 4, p589–599.e5, 5 April 2018
Full Abstract
“Human hippocampal neurogenesis drops sharply in children to undetectable levels in adults.”
Nature volume555, pages377–381 (15 March 2018)
Full Abstract
“Neurons generated in the adult brain are recruited into functional circuits.”
Science 07 Sep 1984: Vol. 225, Issue 4666, pp. 1046–1048
Full Abstract
“De novo generation of neuronal cells from the adult mouse brain.”
PNAS September 15, 1992. 89 (18) 8591–8595
Full Abstract
“Neurogenesis in the adult human hippocampus.”
Nature Medicine volume4, pages1313–1317 (1998)
Full Abstract
“Generation of neurons and astrocytes from isolated cells of the adult mammalian central nervous system.”
Science 27 Mar 1992: Vol. 255, Issue 5052, pp. 1707–1710
Full Abstract
“Autoradiographic and histological evidence of postnatal hippocampal neurogenesis in rats.”
Wiley Online Library First published: June 1965
Full Abstract