. @hjbentham . @clubofinfo . @dissidentvoice_ .#tech .#gmo .#ethics . @ieet .
Tag: medicine

Human Destiny is to Eliminate Death — Essays, Rants & Arguments on Immortalism (Edited Volume)
Immortal Life has complied an edited volume of essays, arguments, and debates about Immortalism titled Human Destiny is to Eliminate Death from many esteemed ImmortalLife.info Authors (a good number of whom are also Lifeboat Foundation Advisory Board members as well), such as Martine Rothblatt (Ph.D, MBA, J.D.), Marios Kyriazis (MD, MS.c, MI.Biol, C.Biol.), Maria Konovalenko (M.Sc.), Mike Perry (Ph.D), Dick Pelletier, Khannea Suntzu, David Kekich (Founder & CEO of MaxLife Foundation), Hank Pellissier (Founder of Immortal Life), Eric Schulke & Franco Cortese (the previous Managing Directors of Immortal Life), Gennady Stolyarov II, Jason Xu (Director of Longevity Party China and Longevity Party Taiwan), Teresa Belcher, Joern Pallensen and more. The anthology was edited by Immortal Life Founder & Senior Editor, Hank Pellissier.
This one-of-a-kind collection features ten debates that originated at ImmortalLife.info, plus 36 articles, essays and diatribes by many of IL’s contributors, on topics from nutrition to mind-filing, from teleomeres to “Deathism”, from libertarian life-extending suggestions to religion’s role in RLE to immortalism as a human rights issue.
The book is illustrated with famous paintings on the subject of aging and death, by artists such as Goya, Picasso, Cezanne, Dali, and numerous others.
The book was designed by Wendy Stolyarov; edited by Hank Pellissier; published by the Center for Transhumanity. This edited volume is the first in a series of quarterly anthologies planned by Immortal Life
Find it on Amazon HERE and on Smashwords HERE
This Immortal Life Anthology includes essays, articles, rants and debates by and between some of the leading voices in Immortalism, Radical Life-Extension, Superlongevity and Anti-Aging Medicine.
A (Partial) List of the Debaters & Essay Contributors:
Martine Rothblatt Ph.D, MBA, J.D. — inventor of satellite radio, founder of Sirius XM and founder of the Terasem Movement, which promotes technological immortality. Dr. Rothblatt is the author of books on gender freedom (Apartheid of Sex, 1995), genomics (Unzipped Genes, 1997) and xenotransplantation (Your Life or Mine, 2003).
Marios Kyriazis MD, MSc, MIBiol, CBiol. founded the British Longevity Society, was the first to address the free-radical theory of aging in a formal mainstream UK medical journal, has authored dozens of books on life-extension and has discussed indefinite longevity in 700 articles, lectures and media appearances globally.
Maria Konovalenko is a molecular biophysicist and the program coordinator for the Science for Life Extension Foundation. She earned her M.Sc. degree in Molecular Biological Physics at the Moscow Institute of Physics and Technology. She is a co-founder of the International Longevity Alliance.
Jason Xu is the director of Longevity Party China and Longevity Party Taiwan, and he was an intern at SENS.
Mike Perry, PhD. has worked for Alcor since 1989 as Care Services Manager. He has authored or contributed to the automated cooldown and perfusion modeling programs. He is a regular contributor to Alcor newsletters. He has been a member of Alcor since 1984.
David A. Kekich, Founder, President & C.E.O Maximum Life Extension Foundation, works to raise funds for life-extension research. He serves as a Board Member of the American Aging Association, Life Extension Buyers’ Club and Alcor Life Extension Foundation Patient Care Trust Fund. He authored Smart, Strong and Sexy at 100?, a how-to book for extreme life extension.
Eric Schulke is the founder of the Movement for Indefinite Life Extension (MILE). He was a Director, Teams Coordinator and ran Marketing & Outreach at the Immortality Institute, now known as Longecity, for 4 years. He is the Co-Managing Director of Immortal Life.
Hank Pellissier is the Founder & Senior Editor of ImmortaLife.info. Previously, he was the founder/director of Transhumanity.net. Before that, he was Managing Director of the Institute for Ethics and Emerging Technology (ieet.org). He’s written over 120 futurist articles for IEET, Hplusmagazine.com, Transhumanity.net, ImmortalLife.info and the World Future Society.
Franco Cortese is on the Advisory Board for Lifeboat Foundation on their Scientific Advisory Board (Life-Extension Sub-Board) and their Futurism Board. He is the Co-Managing Director alongside of Immortal Life and a Staff Editor for Transhumanity. He has written over 40 futurist articles and essays for H+ Magazine, The Institute for Ethics & Emerging Technologies, Immortal Life, Transhumanity and The Rational Argumentator.
Gennady Stolyarov II is a Staff Editor for Transhumanity, Contributor to Enter Stage Right, Le Quebecois Libre, Rebirth of Reason, Ludwig von Mises Institute, Senior Writer for The Liberal Institute, and Editor-in-Chief of The Rational Argumentator.
Brandon King is Co-Director of the United States Longevity Party.
Khannea Suntzu is a transhumanist and virtual activist, and has been covered in articles in Le Monde, CGW and Forbes.
Teresa Belcher is an author, blogger, Buddhist, consultant for anti-aging, life extension, healthy life style and happiness, and owner of Anti-Aging Insights.
Dick Pelletier is a weekly columnist who writes about future science and technologies for numerous publications.
Joern Pallensen has written articles for Transhumanity and the Institute for Ethics and Emerging Technologies.
CONTENTS:
Editor’s Introduction
DEBATES
1. In The Future, With Immortality, Will There Still Be Children?
2. Will Religions promising “Heaven” just Vanish, when Immortality on Earth is attained?
3. In the Future when Humans are Immortal — what will happen to Marriage?
4. Will Immortality Change Prison Sentences? Will Execution and Life-Behind-Bars be… Too Sadistic?
5. Will Government Funding End Death, or will it be Attained by Private Investment?
6. Will “Meatbag” Bodies ever be Immortal? Is “Cyborgization” the only Logical Path?
7. When Immortality is Attained, will People be More — or Less — Interested in Sex?
8. Should Foes of Immortality be Ridiculed as “Deathists” and “Suicidalists”?
9. What’s the Best Strategy to Achieve Indefinite Life Extension?
ESSAYS
1. Maria Konovalenko:
I am an “Aging Fighter” Because Life is the Main Human Right, Demand, and Desire
2. Mike Perry:
Deconstructing Deathism — Answering Objections to Immortality
3. David A. Kekich:
How Old Are You Now?
4. David A. Kekich:
Live Long… and the World Prospers
5. David A. Kekich:
107,000,000,000 — what does this number signify?
6. Franco Cortese:
Religion vs. Radical Longevity: Belief in Heaven is the Biggest Barrier to Eternal Life?!
7. Dick Pelletier:
Stem Cells and Bioprinters Take Aim at Heart Disease, Cancer, Aging
8. Dick Pelletier:
Nanotech to Eliminate Disease, Old Age; Even Poverty
9. Dick Pelletier:
Indefinite Lifespan Possible in 20 Years, Expert Predicts
10. Dick Pelletier:
End of Aging: Life in a World where People no longer Grow Old and Die
11. Eric Schulke:
We Owe Pursuit of Indefinite Life Extension to Our Ancestors
12. Eric Schulke:
Radical Life Extension and the Spirit at the core of a Human Rights Movement
13. Eric Schulke:
MILE: Guide to the Movement for Indefinite Life Extension
14. Gennady Stolyarov II:
The Real War and Why Inter-Human Wars Are a Distraction
15. Gennady Stolyarov II:
The Breakthrough Prize in Life Sciences — turning the tide for life extension
16. Gennady Stolyarov II:
Six Libertarian Reforms to Accelerate Life Extension
17. Hank Pellissier:
Wake Up, Deathists! — You DO Want to LIVE for 10,000 Years!
18. Hank Pellissier:
Top 12 Towns for a Healthy Long Life
19. Hank Pellissier:
This list of 30 Billionaires — Which One Will End Aging and Death?
20. Hank Pellissier:
People Who Don’t Want to Live Forever are Just “Suicidal”
21. Hank Pellissier:
Eluding the Grim Reaper with 23andMe.com
22. Hank Pellissier:
Sixty Years Old — is my future short and messy, or long and glorious?
23. Jason Xu:
The Unstoppable Longevity Virus
24. Joern Pallensen:
Vegetarians Live Longer, Happier Lives
25. Franco Cortese:
Killing Deathist Cliches: Death to “Death-Gives-Meaning-to-Life”
26. Marios Kyriazis:
Environmental Enrichment — Practical Steps Towards Indefinite Lifespans
27. Khannea Suntzu:
Living Forever — the Biggest Fear in the most Audacious Hope
28. Martine Rothblatt:
What is Techno-Immortality?
29. Teresa Belcher:
Top Ten Anti-Aging Supplements
30. Teresa Belcher:
Keep Your Brain Young! — tips on maintaining healthy cognitive function
31. Teresa Belcher:
Anti-Aging Exercise, Diet, and Lifestyle Tips
32. Teresa Belcher:
How Engineered Stem Cells May Enable Youthful Immortality
33. Teresa Belcher:
Nanomedicine — an Introductory Explanation
34. Rich Lee:
“If Eternal Life is a Medical Possibility, I Will Have It Because I Am A Tech Pirate”
35. Franco Cortese:
Morality ==> Immortality
36. Franco Cortese:
Longer Life or Limitless Life?
Intimations of Imitations: Visions of Cellular Prosthesis & Functionally-Restorative Medicine
In this essay I argue that technologies and techniques used and developed in the fields of Synthetic Ion Channels and Ion Channel Reconstitution, which have emerged from the fields of supramolecular chemistry and bio-organic chemistry throughout the past 4 decades, can be applied towards the purpose of gradual cellular (and particularly neuronal) replacement to create a new interdisciplinary field that applies such techniques and technologies towards the goal of the indefinite functional restoration of cellular mechanisms and systems, as opposed to their current proposed use of aiding in the elucidation of cellular mechanisms and their underlying principles, and as biosensors.
In earlier essays (see here and here) I identified approaches to the synthesis of non-biological functional equivalents of neuronal components (i.e. ion-channels ion-pumps and membrane sections) and their sectional integration with the existing biological neuron — a sort of “physical” emulation if you will. It has only recently come to my attention that there is an existing field emerging from supramolecular and bio-organic chemistry centered around the design, synthesis, and incorporation/integration of both synthetic/artificial ion channels and artificial bilipid membranes (i.e. lipid bilayer). The potential uses for such channels commonly listed in the literature have nothing to do with life-extension however, and the field is to my knowledge yet to envision the use of replacing our existing neuronal components as they degrade (or before they are able to), rather seeing such uses as aiding in the elucidation of cellular operations and mechanisms and as biosensors. I argue here that the very technologies and techniques that constitute the field (Synthetic Ion-Channels & Ion-Channel/Membrane Reconstitution) can be used towards the purpose of the indefinite-longevity and life-extension through the iterative replacement of cellular constituents (particularly the components comprising our neurons – ion-channels, ion-pumps, sections of bi-lipid membrane, etc.) so as to negate the molecular degradation they would have otherwise eventually undergone.
While I envisioned an electro-mechanical-systems approach in my earlier essays, the field of Synthetic Ion-Channels from the start in the early 70’s applied a molecular approach to the problem of designing molecular systems that produce certain functions according to their chemical composition or structure. Note that this approach corresponds to (or can be categorized under) the passive-physicalist sub-approach of the physicalist-functionalist approach (the broad approach overlying all varieties of physically-embodied, “prosthetic” neuronal functional replication) identified in an earlier essay.
The field of synthetic ion channels is also referred to as ion-channel reconstitution, which designates “the solubilization of the membrane, the isolation of the channel protein from the other membrane constituents and the reintroduction of that protein into some form of artificial membrane system that facilitates the measurement of channel function,” and more broadly denotes “the [general] study of ion channel function and can be used to describe the incorporation of intact membrane vesicles, including the protein of interest, into artificial membrane systems that allow the properties of the channel to be investigated” [1]. The field has been active since the 1970s, with experimental successes in the incorporation of functioning synthetic ion channels into biological bilipid membranes and artificial membranes dissimilar in molecular composition and structure to biological analogues underlying supramolecular interactions, ion selectivity and permeability throughout the 1980’s, 1990’s and 2000’s. The relevant literature suggests that their proposed use has thus far been limited to the elucidation of ion-channel function and operation, the investigation of their functional and biophysical properties, and in lesser degree for the purpose of “in-vitro sensing devices to detect the presence of physiologically-active substances including antiseptics, antibiotics, neurotransmitters, and others” through the “… transduction of bioelectrical and biochemical events into measurable electrical signals” [2].
Thus my proposal of gradually integrating artificial ion-channels and/or artificial membrane sections for the purpse of indefinite longevity (that is, their use in replacing existing biological neurons towards the aim of gradual substrate replacement, or indeed even in the alternative use of constructing artificial neurons to, rather than replace existing biological neurons, become integrated with existing biological neural networks towards the aim of intelligence amplification and augmentation while assuming functional and experiential continuity with our existing biological nervous system) appears to be novel, while the notion of artificial ion-channels and neuronal membrane systems ion general had already been conceived (and successfully created/experimentally-verified, though presumably not integrated in-vivo).
The field of Functionally-Restorative Medicine (and the orphan sub-field of whole-brain-gradual-substrate-replacement, or “physically-embodied” brain-emulation if you like) can take advantage of the decades of experimental progress in this field, incorporating both the technological and methodological infrastructures used in and underlying the field of Ion-Channel Reconstitution and Synthetic/Artificial Ion Channels & Membrane-Systems (and the technologies and methodologies underlying their corresponding experimental-verification and incorporation techniques) for the purpose of indefinite functional restoration via the gradual and iterative replacement of neuronal components (including sections of bilipid membrane, ion channels and ion pumps) by MEMS (micro-electrocal-mechanical-systems) or more likely NEMS (nano-electro-mechanical systems).
The technological and methodological infrastructure underlying this field can be utilized for both the creation of artificial neurons and for the artificial synthesis of normative biological neurons. Much work in the field required artificially synthesizing cellular components (e.g. bilipid membranes) with structural and functional properties as similar to normative biological cells as possible, so that the alternative designs (i.e. dissimilar to the normal structural and functional modalities of biological cells or cellular components) and how they affect and elucidate cellular properties, could be effectively tested. The iterative replacement of either single neurons, or the sectional replacement of neurons with synthesized cellular components (including sections of the bi-lipid membrane, voltage-dependent ion-channels, ligand-dependent ion channels, ion pumps, etc.) is made possible by the large body of work already done in the field. Consequently the technological, methodological and experimental infrastructures developed for the fields of Synthetic
Ion-Channels and Ion-Channel/Artificial-Membrane-Reconstitution can be utilized for the purpose of a.) iterative replacement and cellular upkeep via biological analogues (or not differing significantly in structure or functional & operational modality to their normal biological counterparts) and/or b.) iterative replacement with non-biological analogues of alternate structural and/or functional modalities.
Rather than sensing when a given component degrades and then replacing it with an artificially-synthesized biological or non-biological analogue, it appears to be much more efficient to determine the projected time it takes for a given component to degrade or otherwise lose functionality, and simply automate the iterative replacement in this fashion, without providing in-vivo systems for detecting molecular or structural degradation. This would allow us to achieve both experimental and pragmatic success in such cellular-prosthesis sooner, because it doesn’t rely on the complex technological and methodological infrastructure underlying in-vivo sensing, especially on the scale of single neuron components like ion-channels, and without causing operational or functional distortion to the components being sensed.
A survey of progress in the field [3] lists several broad design motifs. I will first list the deign motifs falling within the scope of the survey, and the examples it provides. Selections from both papers are meant to show the depth and breadth of the field, rather than to elucidate the specific chemical or kinetic operations under the purview of each design-variety.
For a much more comprehensive, interactive bibliography of papers falling within the field of Synthetic Ion-Channels or constituting the historical foundations of the field, see Jon Chui’s online biography here, which charts the developments in this field up until 2011.
First Survey
Unimolecular ion channels:
Examples include a.) synthetic ion channels with oligocrown ionophores, [5] b.) using a-helical peptide scaffolds and rigid push–pull p-octiphenyl scaffolds for the recognition of polarized membranes, [6] and c.) modified varieties of the b-helical scaffold of gramicidin A [7]
Barrel-stave supramolecules:
Examples of this general class falling include avoltage-gated synthetic ion channels formed by macrocyclic bolaamphiphiles and rigidrod p-octiphenyl polyols [8].
Macrocyclic, branched and linear non-peptide bolaamphiphiles as staves:
Examples of this sub-class include synthetic ion channels formed by a.) macrocyclic, branched and linear bolaamphiphiles and dimeric steroids, [9] and by b.) non-peptide macrocycles, acyclic analogs and peptide macrocycles [respectively] containing abiotic amino acids [10].
Dimeric steroid staves:
Examples of this sub-class include channels using polydroxylated norcholentriol dimer [11].
pOligophenyls as staves in rigid rod b barrels:
Examples of this sub-class include “cylindrical self-assembly of rigid-rod b-barrel pores preorganized by the nonplanarity of p-octiphenyl staves in octapeptide-p-octiphenyl monomers” [12].
Synthetic Polymers:
Examples of this sub-class include synthetic ion channels and pores comprised of a.) polyalanine, b.) polyisocyanates, c.) polyacrylates, [13] formed by i.) ionophoric, ii.) ‘smart’ and iii.) cationic polymers [14]; d.) surface-attached poly(vinyl-n-alkylpyridinium) [15]; e.) cationic oligo-polymers [16] and f.) poly(m-phenylene ethylenes) [17].
Helical b-peptides (used as staves in barrel-stave method):
Examples of this class include: a.) cationic b-peptides with antibiotic activity, presumably acting as amphiphilic helices that form micellar pores in anionic bilayer membranes [18].
Monomeric steroids:
Examples of this sub-class falling include synthetic carriers, channels and pores formed by monomeric steroids [19], synthetic cationic steroid antibiotics [that] may act by forming micellar pores in anionic membranes [20], neutral steroids as anion carriers [21] and supramolecular ion channels [22].
Complex minimalist systems:
Examples of this sub-class falling within the scope of this survey include ‘minimalist’ amphiphiles as synthetic ion channels and pores [23], membrane-active ‘smart’ double-chain amphiphiles, expected to form ‘micellar pores’ or self-assemble into ion channels in response to acid or light [24], and double-chain amphiphiles that may form ‘micellar pores’ at the boundary between photopolymerized and host bilayer domains and representative peptide conjugates that may self assemble into supramolecular pores or exhibit antibiotic activity [25].
Non-peptide macrocycles as hoops:
Examples of this sub-class falling within the scope of this survey include synthetic ion channels formed by non-peptide macrocycles acyclic analogs [26] and peptide macrocycles containing abiotic amino acids [27].
Peptide macrocycles as hoops and staves:
Examples of this sub-class include a.) synthetic ion channels formed by self-assembly of macrocyclic peptides into genuine barrel-hoop motifs that mimic the b-helix of gramicidin A with cyclic b-sheets. The macrocycles are designed to bind on top of channels and cationic antibiotics (and several analogs) are proposed to form micellar pores in anionic membranes [28]; b.) synthetic carriers, antibiotics (and analogs) and pores (and analogs) formed by macrocyclic peptides with non-natural subunits. [Certain] macrocycles may act as b-sheets, possibly as staves of b-barrel-like pores [29]; c.) bioengineered pores as sensors. Covalent capturing and fragmentations [have been] observed on the single-molecule level within engineered a-hemolysin pore containing an internal reactive thiol [30].
Summary
Thus even without knowledge of supramolecular or organic chemistry, one can see that a variety of alternate approaches to the creation of synthetic ion channels, and several sub-approaches within each larger ‘design motif’ or broad-approach, not only exist but have been experimentally verified, varietized and refined.
Second Survey
The following selections [31] illustrate the chemical, structural and functional varieties of synthetic ions categorized according to whether they are cation-conducting or anion-conducting, respectively. These examples are used to further emphasize the extent of the field, and the number of alternative approaches to synthetic ion-channel design, implementation, integration and experimental-verification already existent. Permission to use all the following selections and figures were obtained from the author of the source.
There are 6 classical design-motifs for synthetic ion-channels, categorized by structure, that are identified within the paper:
A: unimolecular macromolecules,
B: complex barrel-stave,
C: barrel-rosette,
D: barrel hoop, and
E: micellar supramolecules.
Cation Conducting Channels:
UNIMOLECULAR
“The first non-peptidic artificial ion channel was reported by Kobuke et al. in 1992” [33].
“The channel contained “an amphiphilic ion pair consisting of oligoether-carboxylates and mono- (or di-) octadecylammoniumcations. The carboxylates formed the channel core and the cations formed the hydrophobic outer wall, which was embedded in the bilipid membrane with a channel length of about 24 to 30 Å. The resultant ion channel, formed from molecular self-assembly, is cation selective and voltage-dependent” [34].
“Later, Kokube et al. synthesized another channel comprising of resorcinol based cyclic tetramer as the building block. The resorcin-[4]-arenemonomer consisted of four long alkyl chains which aggregated to forma dimeric supramolecular structure resembling that of Gramicidin A” [35]. “Gokel et al. had studied [a set of] simple yet fully functional ion channels known as “hydraphiles” [39].
“An example (channel 3) is shown in Figure 1.6, consisting of diaza-18-crown-6 crown ether groups and alkyl chain as side arms and spacers. Channel 3 is capable of transporting protons across the bilayer membrane” [40].
“A covalently bonded macrotetracycle4 (Figure 1.8) had shown to be about three times more active than Gokel’s ‘hydraphile’ channel, and its amide-containing analogue also showed enhanced activity” [44].
“Inorganic derivative using crown ethers have also been synthesized. Hall et. al synthesized an ion channel consisting of a ferrocene and 4 diaza-18-crown-6 linked by 2 dodecyl chains (Figure 1.9). The ion channel was redox-active as oxidation of the ferrocene caused the compound to switch to an inactive form” [45]
B STAVES:
“These are more difficult to synthesize [in comparison to unimolecular varieties] because the channel formation usually involves self-assembly via non-covalent interactions” [47].“A cyclic peptide composed of even number of alternating D- and L-amino acids (Figure 1.10) was suggested to form barrel-hoop structure through backbone-backbone hydrogen bonds by De Santis” [49].
“A tubular nanotube synthesized by Ghadiri et al. consisting of cyclic D and L peptide subunits form a flat, ring-shaped conformation that stack through an extensive anti-parallel β-sheet-like hydrogen bonding interaction (Figure 1.11)” [51].
“Experimental results have shown that the channel can transport sodium and potassium ions. The channel can also be constructed by the use of direct covalent bonding between the sheets so as to increase the thermodynamic and kinetic stability” [52].
“By attaching peptides to the octiphenyl scaffold, a β-barrel can be formed via self-assembly through the formation of β-sheet structures between the peptide chains (Figure 1.13)” [53].
“The same scaffold was used by Matile etal. to mimic the structure of macrolide antibiotic amphotericin B. The channel synthesized was shown to transport cations across the membrane” [54].
“Attaching the electron-poor naphthalenediimide (NDIs) to the same octiphenyl scaffold led to the hoop-stave mismatch during self-assembly that results in a twisted and closed channel conformation (Figure 1.14). Adding the compleentary dialkoxynaphthalene (DAN) donor led to the cooperative interactions between NDI and DAN that favors the formation of barrel-stave ion channel.” [57].
MICELLAR
“These aggregate channels are formed by amphotericin involving both sterols and antibiotics arranged in two half-channel sections within the membrane” [58].
“An active form of the compound is the bolaamphiphiles (two-headed amphiphiles). (Figure 1.15) shows an example that forms an active channel structure through dimerization or trimerization within the bilayer membrane. Electrochemical studies had shown that the monomer is inactive and the active form involves dimer or larger aggregates” [60].
ANION CONDUCTING CHANNELS:
“A highly active, anion selective, monomeric cyclodextrin-based ion channel was designed by Madhavan et al (Figure 1.16). Oligoether chains were attached to the primary face of the β-cyclodextrin head group via amide bonds. The hydrophobic oligoether chains were chosen because they are long enough to span the entire lipid bilayer. The channel was able to select “anions over cations” and “discriminate among halide anions in the order I-> Br-> Cl- (following Hofmeister series)” [61].
“The anion selectivity occurred via the ring of ammonium cations being positioned just beside the cyclodextrin head group, which helped to facilitate anion selectivity. Iodide ions were transported the fastest because the activation barrier to enter the hydrophobic channel core is lower for I- compared to either Br- or Cl-“ [62]. “A more specific artificial anion selective ion channel was the chloride selective ion channel synthesized by Gokel. The building block involved a heptapeptide with Proline incorporated (Figure 1.17)” [63].
Cellular Prosthesis: Inklings of a New Interdisciplinary Approach
The paper cites “nanoreactors for catalysis and chemical or biological sensors” and “interdisciplinary uses as nano –filtration membrane, drug or gene delivery vehicles/transporters as well as channel-based antibiotics that may kill bacterial cells preferentially over mammalian cells” as some of the main applications of synthetic ion-channels [65], other than their normative use in elucidating cellular function and operation.
However, I argue that a whole interdisciplinary field and heretofore-unrecognized new approach or sub-field of Functionally-Restorative Medicine is possible through taking the technologies and techniques involved in in constructing, integrating, and experimentally-verifying either a.) non-biological analogues of ion-channels & ion-pumps (thus trans-membrane membrane proteins in general, also sometimes referred to as transport proteins or integral membrane proteins) and membranes (which include normative bilipid membranes, non-lipid membranes and chemically-augmented bilipid membranes), and b.) the artificial synthesis of biological analogues of ion-channels, ion-pumps and membranes, which are structurally and chemically equivalent to naturally-occurring biological components but which are synthesized artificially – and applying such technologies and techniques toward the purpose the gradual replacement of our existing biological neurons constituting our nervous systems – or at least those neuron-populations that comprise the neo- and prefrontal-cortex, and through iterative procedures of gradual replacement thereby achieving indefinite-longevity. There is still work to be done in determining the comparative advantages and disadvantages of various structural and functional (i.e. design) motifs, and in the logistics of implanting the iterative replacement or reconstitution of ion-channels, ion-pumps and sections of neuronal membrane in-vivo.
The conceptual schemes outlined in Concepts for Functional Replication of Biological Neurons [66], Gradual Neuron Replacement for the Preservation of Subjective-Continuity [67] and Wireless Synapses, Artificial Plasticity, and Neuromodulation [68] would constitute variations on the basic approach underlying this proposed, embryonic interdisciplinary field. Certain approaches within the fields of nanomedicine itself, particularly those approaches that constitute the functional emulation of existing cell-types, such as but not limited to Robert Freitas’s conceptual designs for the functional emulation of the red blood cell (a.k.a. erythrocytes, haematids) [69], i.e. the Resperocyte, itself should be seen as falling under the purview of this new approach, although not all approaches to Nanomedicine (diagnostics, drug-delivery and neuroelectronic interfacing) constitute the physical (i.e. electromechanical, kinetic and/or molecular physically-embodied) and functional emulation of biological cells.
The field of functionally-restorative medicine in general (and of nanomedicine in particular) and the field of supramolecular and organic chemistry converge here, where these technological, methodological, and experimental infrastructures developed in field of Synthetic Ion-Channels and Ion Channel Reconstitution can be employed to develop a new interdisciplinary approach that applies the logic of prosthesis to the cellular and cellular-component (i.e. sub-cellular) scale; same tools, new use. These techniques could be used to iteratively replace the components of our neurons as they degrade, or to replace them with more robust systems that are less susceptible to molecular degradation. Instead of repairing the cellular DNA, RNA and protein transcription and synthesis machinery, we bypass it completely by configuring and integrating the neuronal components (ion-channels, ion-pumps and sections of bilipid membrane) directly.
Thus I suggest that theoreticians of nanomedicine look to the large quantity of literature already developed in the emerging fields of synthetic ion-channels and membrane-reconstitution, towards the objective of adapting and applying existing technologies and methodologies to the new purpose of iterative maintenance, upkeep and/or replacement of cellular (and particularly neuronal) constituents with either non-biological analogues or artificially-synthesized-but-chemically/structurally-equivalent biological analogues.
This new sub-field of Synthetic Biology needs a name to differentiate it from the other approaches to Functionally-Restorative Medicine. I suggest the designation ‘cellular prosthesis’.
References:
[1] Williams (1994)., An introduction to the methods available for ion channel reconstitution. in D.C Ogden Microelectrode techniques, The Plymouth workshop edition, CambridgeCompany of Biologists.
[2] Tomich, J., Montal, M. (1996). U.S Patent No. 5,16,890. Washington, DC: U.S. Patent and Trademark Office.
[3] Matile, S., Som, A., & Sorde, N. (2004). Recent synthetic ion channels and pores. Tetrahedron, 60(31), 6405-6435. ISSN 0040-4020, 10.1016/j.tet.2004.05.052. Access: http://www.sciencedirect.com/science/article/pii/S0040402004007690:
[4] XIAO, F., (2009). Synthesis and structural investigations of pyridine-based aromatic foldamers.
[5] Ibid., p. 6411.
[6] Ibid., p. 6416.
[7] Ibid., p. 6413.
[8] Ibid., p. 6412.
[9] Ibid., p. 6414.
[10] Ibid., p. 6425.
[11] Ibid., p. 6427.
[12] Ibid., p. 6416.
[13] Ibid., p. 6419.
[14] Ibid., p. 6419.
[15] Ibid., p. 6419.
[16] Ibid., p. 6419.
[17] Ibid., p. 6419.
[18] Ibid., p. 6421.
[19] Ibid., p. 6422.
[20] Ibid., p. 6422.
[21] Ibid., p. 6422.
[22] Ibid., p. 6422.
[23] Ibid., p. 6423.
[24] Ibid., p. 6423.
[25] Ibid., p. 6423.
[26] Ibid., p. 6426.
[27] Ibid., p. 6426.
[28] Ibid., p. 6427.
[29] Ibid., p. 6327.
[30] Ibid., p. 6427.
[31] XIAO, F. (2009). Synthesis and structural investigations of pyridine-based aromatic foldamers.
[32] Ibid., p. 4.
[33] Ibid., p. 4.
[34] Ibid., p. 4.
[35] Ibid., p. 4.
[36] Ibid., p. 7.
[37] Ibid., p. 8.
[38] Ibid., p. 7.
[39] Ibid., p. 7.
[40] Ibid., p. 7.
[41] Ibid., p. 7.
[42] Ibid., p. 7.
[43] Ibid., p. 8.
[44] Ibid., p. 8.
[45] Ibid., p. 9.
[46] Ibid., p. 9.
[47] Ibid., p. 9.
[48] Ibid., p. 10.
[49] Ibid., p. 10.
[50] Ibid., p. 10.
[51] Ibid., p. 10.
[52] Ibid., p. 11.
[53] Ibid., p. 12.
[54] Ibid., p. 12.
[55] Ibid., p. 12.
[56] Ibid., p. 12.
[57] Ibid., p. 12.
[58] Ibid., p. 13.
[59] Ibid., p. 13.
[60] Ibid., p. 14.
[61] Ibid., p. 14.
[62] Ibid., p. 14.
[63] Ibid., p. 15.
[64] Ibid., p. 15.
[65] Ibid., p. 15.
[66] Cortese, F., (2013). Concepts for Functional Replication of Biological Neurons. The Rational Argumentator. Access: http://www.rationalargumentator.com/index/blog/2013/05/concepts-functional-replication/
[67] Cortese, F., (2013). Gradual Neuron Replacement for the Preservation of Subjective-Continuity. The Rational Argumentator. Access: http://www.rationalargumentator.com/index/blog/2013/05/gradual-neuron-replacement/
[68] Cortese, F., (2013). Wireless Synapses, Artificial Plasticity, and Neuromodulation. The Rational Argumentator. Access: http://www.rationalargumentator.com/index/blog/2013/05/wireless-synapses/
[69] Freitas Jr., R., (1998). “Exploratory Design in Medical Nanotechnology: A Mechanical Artificial Red Cell”. Artificial Cells, Blood Substitutes, and Immobil. Biotech. (26): 411–430. Access: http://www.ncbi.nlm.nih.gov/pubmed/9663339
If you want to live longer, do nothing
By Avi Roy, University of Buckingham
I want to live longer and help others do the same. I assumed the most effective way to do that is by understanding the science of aging and then engineering solutions to extend human lifespan. That is why I became a biomedical researcher and over the past several years I have pursued this goal almost single-mindedly.
When a 2004 study showed that reducing the calorie intake in mice extended their life by 42%, I enthusiastically embraced the results and even put myself on a calorie restricted diet. But, subsequently, a 2012 study showed that long-term calorie restriction may not have the promised benefits. On the contrary, fewer calories without the required nutrients might actually cause harm.
Calorie restriction is not the first such “promising” route that eventually did not live up to the promise, and it will not be the last. Antioxidants showed promise in holding back diseases caused by aging, but now we know that antioxidant supplements are more likely to shorten your life.
Earlier in May, researchers showed that reducing a protein called NF-kB in mouse brains modestly improved their lifespan. I am not holding out for this result either. Before too long, I’m sure there will be reports of severe side effects of manipulating levels of NF-kB.
Take it easy
Looking at the data I have come to the conclusion that “doing nothing” may be the best option in most cases. This may not be as pessimistic as it sounds and it is definitely not to say that research in fighting aging must not be carried out.
When I say “do nothing”, I am assuming that you do not smoke or drink too much alcohol, and have access to medical care in case of injury. Such measures are bound to increase your lifespan.
But currently, not intervening in the aging process is more likely to help you live longer than trying any of the methods I’ve mentioned, not by a few months but by many years. Trying any of those interventions may actually cause harm, and will do so for the foreseeable future.
Lesson from the past
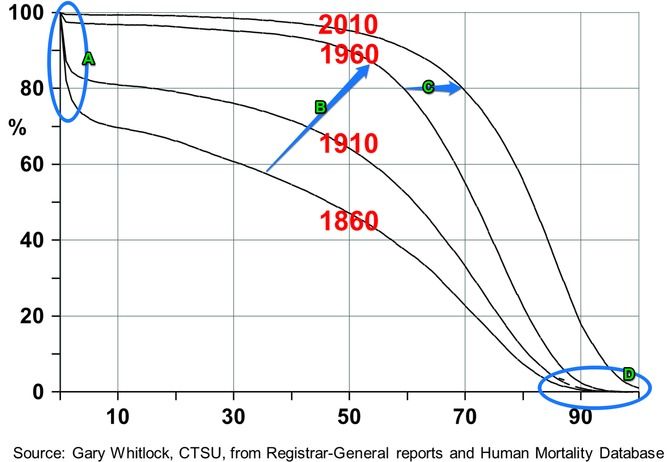
The chart below shows the survival rates – the percentage of the population that lives to a certain age – for men in England and Wales from 1860 to 2010.
In the 1860s, more than 20% of children died at birth or soon after. On average, men’s health started to decline around the age of 30, and only about 20% of the population survived for more than 70 years.
By 1910, child mortality decreased, thanks to improvements in hygiene and better medical care. This meant more men lived past the age of 50. Circle A shows this reduction in childhood mortality between 1860 and 2010. But, as can be seen from Circle D, the gain towards the end was not significant. This is because only 30% of males passed the age of 70.
Fifty years later, after the discovery of penicillin and invention of more vaccines, 90% of English and Welsh men lived until 50, and more than half survived to 70. Arrow B marks this trend.
Today almost 80% of men live to the age of 70. Four times as many men reach 70 now than in 1860.
What accounts for the change? Between 1860 and 1960, the significant increase in survival rate was due to medical intervention. Since 1960, the survival curve has improved mainly due to reduction in smoking.
This trend is similar in many rich countries, including the US. Druin Burch, a physician and writer, says in his book Taking the Medicine, that eliminating smoking would provide more benefits than being able to cure people of every possible type of cancer.
Age gracefully
Many experts believe that human lifespan might actually have an upper limit of 125 years. The average may not increase much beyond 90. If we are to agree with them, this leaves little room for improvement.
But we have never concentrated on maximising human lifespan before. Most people believe human lifespan is finite, so all drugs being manufactured today are targeted towards certain age-related diseases such as diabetes and hypertension. They are not designed to extend human lifespan.
If this bleak outlook is indeed true, we should not practise naive interventionism because it is unlikely to help. As Nassim Nicholas Taleb describes in his book Antifragile, naive interventionism occurs when we try to fix a single thing, but end up disturbing a complex system.
In case of extending human lifespan, those naive interventions would include calorie restriction, antioxidant supplements or manipulating the protein NF-kB, as mentioned earlier. They also include the current obsession with replacing fat in foods with sugar, the health benefits of drinking red wine, or the use ofsurgery or supplements to “fight” aging. This latter industry has grown in the past decade from being non-existent to an estimated worth of $88 billion today.
If intervening in the aging process with current biomedical science has any positive effect at all, it will be far too small to worry about. It’s far more likely to harm us.
That is why I have decided to do nothing and follow a simple rule: unless I meet with an accident, or suffer from a terminal disease, I will not add anything to my life with the explicit purpose of extending it. To do anything else would most likely do more harm than good.
Avi Roy does not work for, consult to, own shares in or receive funding from any company or organisation that would benefit from this article, and has no relevant affiliations.
This article was originally published at The Conversation.
Read the original article.