Here, we demonstrated that intradermal administration of clinically relevant vaccines efficiently induces Trm cells specific for tumor-specific and self-antigens that accumulate in vaccinated and non-vaccinated skin. Interestingly, vaccination-induced Trm cells strongly suppress the growth of melanoma, independently of circulating CD8 T cells, and were able to infiltrate melanoma tumors. Therefore, our work highlights the therapeutic potential of vaccination-induced Trm cells to achieve potent protection against skin malignancies.
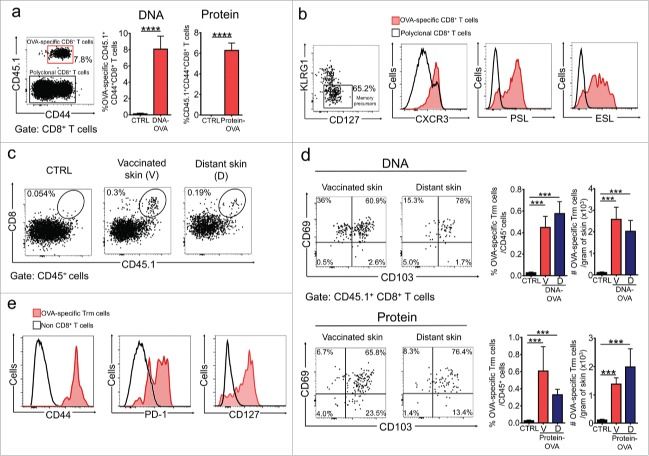
Memory CD8+ T cell responses have the potential to mediate long-lasting protection against cancers. Resident memory CD8+ T (Trm) cells stably reside in non-lymphoid tissues and mediate superior innate and adaptive immunity against pathogens. Emerging evidence indicates that Trm cells develop in human solid cancers and play a key role in controlling tumor growth. However, the specific contribution of Trm cells to anti-tumor immunity is incompletely understood. Moreover, clinically applicable vaccination strategies that efficiently establish Trm cell responses remain largely unexplored and are expected to strongly protect against tumors. Here we demonstrated that a single intradermal administration of gene- or protein-based vaccines efficiently induces specific Trm cell responses against models of tumor-specific and self-antigens, which accumulated in vaccinated and distant non-vaccinated skin. Vaccination-induced Trm cells were largely resistant to in vivo intravascular staining and antibody-dependent depletion. Intradermal, but not intraperitoneal vaccination, generated memory precursors expressing skin-homing molecules in circulation and Trm cells in skin. Interestingly, vaccination-induced Trm cell responses strongly suppressed the growth of B16F10 melanoma, independently of circulating memory CD8+ T cells, and were able to infiltrate tumors. This work highlights the therapeutic potential of vaccination-induced Trm cell responses to achieve potent protection against skin malignancies.
KEYWORDS: Cancer vaccines, DNA vaccines, intradermal vaccination, melanoma, models of anticancer vaccination, protein vaccines, tissue resident memory CD8+ T cells.
Immunotherapy is emerging as a new form to treat cancer by harnessing the activity of cytotoxic CD8+ T lymphocytes (CTLs) that specifically recognize tumor-associated antigens. Transfusion of autologous tumor-specific CTLs1–4 and blockade of T cell inhibitory receptors5–7 have demonstrated to elicit durable clinical benefit in a significant proportion of patients with melanoma, leukemia, lymphoma and other cancers, who failed to respond to conventional treatments. Vaccination strategies eliciting CTL responses specific for tumor-specific and self-antigens have shown promising results in recent clinical trials.8–10 Long-lasting protective immunity relies on the efficient establishment of long-lived memory CD8+ T cells, which have the potential to eradicate primary and disseminated tumors.11 They have been typically classified in two subsets: effector-memory (Tem) and central-memory (Tcm) CD8+ T cells.